HEOR Research in Life Sciences
HEOR Solutions Designed for Modern Evidence GenerationMadeAi transforms how HEOR teams discover, synthesize, and operationalize evidence. Built as a truly AI-native platform, MadeAi accelerates every step of the literature review process—protocol creation, screening, data extraction, quality appraisal, complex summarization, and structured reporting—while preserving the rigor and defensibility HEOR demands.






Discover Proven Results From Our Customer Success Stories
60%
Time Savings
90+%
Accuracy
100%
Client Satisfaction
96+%
Traceability
Powering HEOR Research in Life Sciences while Enhancing Workflow
MadeAi™ transforms by leveraging AI for accelerating evidence synthesis in HEOR. It helps HEOR teams discover, synthesize, and operationalize evidence. Built as a truly AI-native platform, MadeAi accelerates every step of the HEOR process.
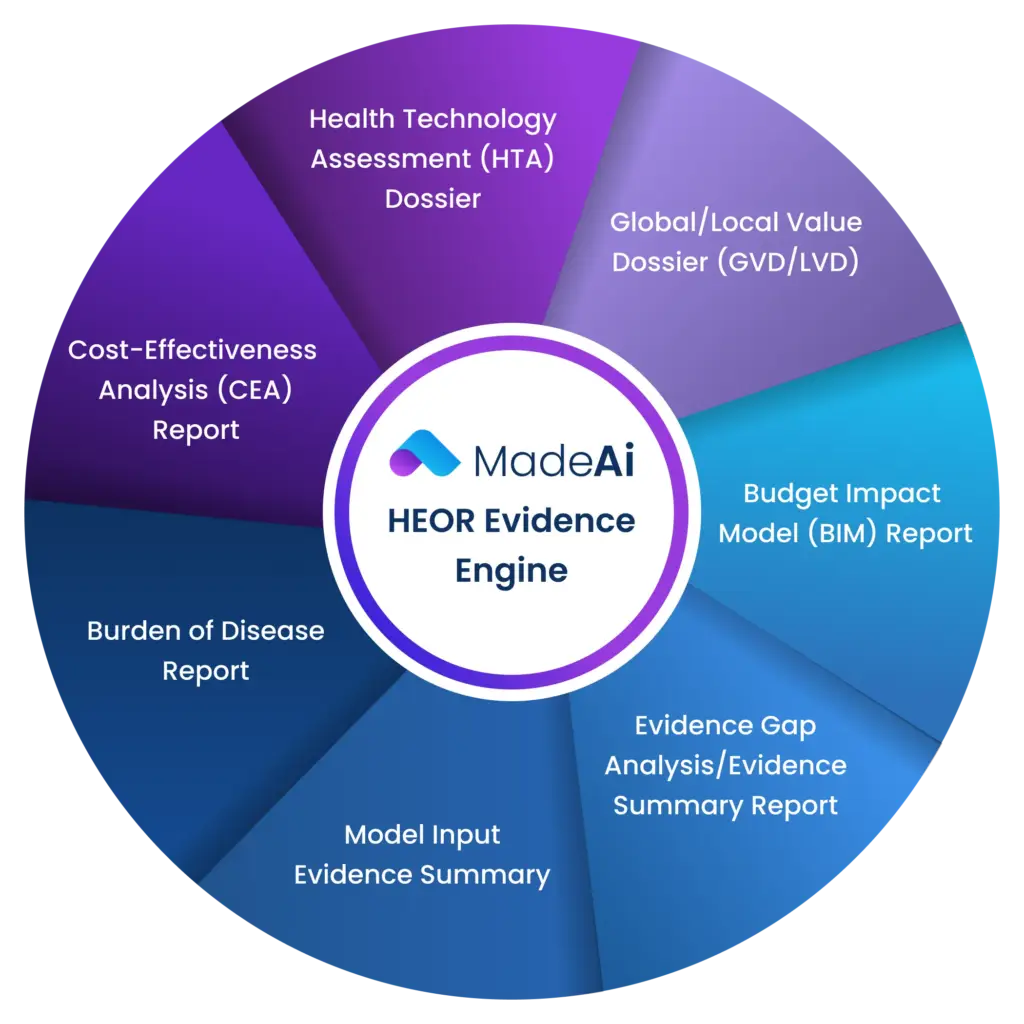
Health Technology Assessment (HTA) Dossier
Supporting reimbursement and access decisions with structured clinical, economic, and comparative evidence aligned to HTA requirements.
Global/Local Value Dossier (GVD/LVD)
Delivering tailored value narratives with clinical, economic, and real-world evidence for global and country-specific submissions.
Budget Impact Model (BIM) Report
Estimating the financial impact of new therapies on healthcare systems, supporting payer decision-making, and planning.
Evidence Gap Analysis/
Evidence Summary Report
Identifying evidence gaps and synthesizing available data to strengthen value communication and support strategic planning.
Model Input Evidence Summary
Aggregating and validating evidence inputs required for economic models, ensuring transparency and traceability.
Burden of Disease Report
Analyzing epidemiology, clinical outcomes, and economic burden to contextualize disease impact and unmet needs.
Cost-Effectiveness Analysis (CEA) Report
Evaluating clinical and economic value of interventions to support pricing, reimbursement, and HTA submissions.
Accelerate Time to Insight
Cut screening, extraction, and reporting time by up-to 60%, enabling the HEOR team to support more studies, more submissions, and more payer deliverables without increasing headcount.
Strengthen Rigor and Consistency
Standardized protocols, guided methodologies, and transparent decision logs ensure reproducible evidence synthesis suitable for HTA and internal governance.
Improve Quality of Economic Inputs
MadeAi’s structured extraction and ARM-level capture of effect sizes, utilities, resource use, safety, and QoL variables feeds economic models with cleaner, more complete data.
Reimbursement Dossier
Supporting payer access, including clinical, economic, and patient outcomes data.
Budget Impact Analyses
Estimating financial impact of product adoption for healthcare systems.
JCA Scoping & Dossier
Sharing an evidence assessment for EU-level HTA requirements including comparators, outcomes, and clinical benefit positioning.
Value Dossier
Providing a comprehensive summary of clinical, economic, and patient value for payers, HTAs, and stakeholders.
Market Access Reports
Summarizing payer policies, guidelines, and market trends impacting product access.
Patient Access Program Reports
Tracking outcomes and effectiveness of patient support programs.
Pricing Strategy Reports
Supporting pricing decisions with clinical, economic, and market evidence.
Scale Evidence Generation Without Bottlenecks
HEOR teams frequently juggle treatment landscape reviews, BOI studies, safety analyses, and model-input searches simultaneously. MadeAi’s AI-driven workflows allow teams to run multiple reviews in parallel.
Enhance Collaboration Across HEOR, MA, Market Access, and RWE
Shared workspaces, consistent tagging schemas, and version-controlled outputs reduce duplication and ensure a unified evidence story across departments.
Submission-Ready Outputs
These HEOR services generate structured PRISMA documentation, evidence tables, summaries of findings, and export packages aligned to HTA standards.
“MadeAi-LR dramatically accelerated our literature review process. What typically takes weeks now takes us just a couple of days. Even more impressive, the platform delivers consistently accurate results that our reviewers trust.”
HEOR Professional
MadeAi User, Pharmaceutical Manufacturer
Case Studies & Scientific Research
Choose the Right Engagement Model
MadeAi Quick Research SaaS Subscription
- Want your internal team to manage research independently?
- Need a cost-effective solution with minimal reliance on external expertise?
AI-Enabled Research Services
- Need experts to handle your quick research?
- Prefer minimal hands-on involvement?
- Short on internal resources or time?
- Working on a critical publication or submission?
Build–Operate–Transfer
- Looking for a partner to help you customize your solution and build adoption internally?
- Need help operationalizing your research program?
- Trying to build a fully custom AI-driven research platform tailored to your organization?
Transform Your HEOR Workflows with AI-Driven Evidence Synthesis
Discover how MadeAi helps streamline HEOR workflows with structured, evidence-driven support.

FAQ
AI-powered evidence synthesis uses artificial intelligence to automate literature review, data extraction, and structured reporting. It enables HEOR teams to analyze large volumes of clinical and economic data faster while maintaining rigor for HTA and market access.
MadeAi for HEOR teams is an AI-native platform that accelerates evidence synthesis across the full literature review workflow, from protocol creation and screening to data extraction, quality appraisal, and structured reporting. It is purpose-built for Health Economics and Outcomes Research (HEOR) professionals who need faster, more transparent, and regulatory-grade outputs for HTA submissions and Real-World Evidence (RWE) generation.
MadeAi reduces screening, extraction, and reporting time by up to 60% by automating repetitive steps while keeping human oversight. HEOR teams can run multiple reviews in parallel, move from protocol to submission-ready outputs faster, and eliminate bottlenecks without sacrificing methodological rigor.
HEOR teams typically save 60% on literature review timelines, turning weeks of work into a couple of days. The platform handles protocol design, screening, extraction, and reporting in one traceable workflow, freeing experts for higher-value analysis and more frequent submissions.
Traceability ensures every decision, source, and data point is auditable and reproducible—critical for HTA bodies and internal governance. MadeAi delivers 96% traceability, giving HEOR, Medical Affairs, and Market Access teams complete confidence in their evidence for Pricing and Reimbursement decisions.
MadeAi uses structured extraction at the ARM level to capture effect sizes, utilities, resource use, safety, and QoL variables with higher precision. This cleaner, more complete data directly feeds into economic models, reducing manual errors and strengthening the robustness of cost-effectiveness analyses.
Submission-ready outputs include PRISMA-aligned documentation, evidence tables, structured summaries of findings, and export packages specifically formatted for HTA requirements. HEOR teams receive auditable, publication-quality materials that accelerate regulatory and payer submissions.
MadeAi offers shared workspaces, consistent tagging schemes, and version-controlled outputs that align evidence across HEOR, Medical Affairs, and Market Access. This unified evidence story reduces duplication and strengthens value dossiers and Pricing and Reimbursement strategies.



