For the last several years, artificial intelligence in life sciences has largely lived in innovation labs and pilot programs—valuable but often disconnected from the workflows that actually drive regulatory approvals, market access, and clinical decision-making. In 2026, that changes.
Our first prediction from MadeAi & CapeStart’s 2026 outlook, developed with insights from Angeline Dhas, Head of Product Management at MadeAi, and Siva Karthick, Machine Learning Lead at MadeAi, signals a clear inflection point: AI is transitioning from experimentation to enterprise-scale production.
From Pilot Projects to Production Systems
Life sciences organizations are no longer asking, “Can AI help?” They’re asking, “Where can AI drive measurable impact?”
Across the industry, AI is being embedded directly into core operational workflows, including:
- R&D evidence landscaping and study design
- Systematic literature reviews and evidence synthesis
- Health technology assessment (HTA) and regulatory submissions
- Medical affairs insights and publication planning
- Market access strategy and value communication
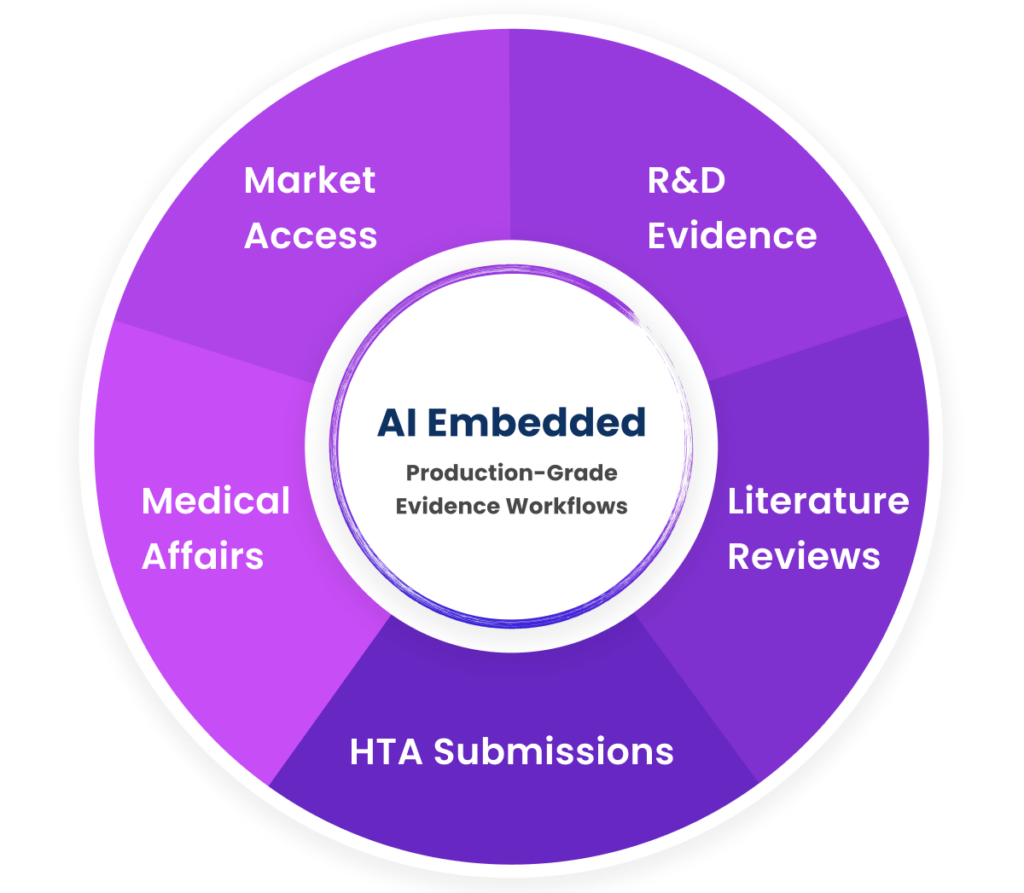
AI in production across the full evidence lifecycle.
This shift reflects a broader maturation of AI. Organizations are moving beyond novelty use cases toward repeatable, validated, and auditable systems that can withstand regulatory scrutiny and internal governance.
The Rise of ROI-Driven AI
In 2026, AI success won’t be measured by adoption alone—operational and financial outcomes will measure it.
Life sciences leaders are increasingly evaluating AI initiatives based on:
- Time savings across evidence generation workflows
- Cost reductions in outsourced research and analysis
- Accuracy and reproducibility of outputs
- Traceability and transparency for regulatory acceptance
- Scalability across global teams and therapeutic areas

Key AI benefits driving the shift from pilots to production systems.
This ROI-driven mindset is a clear departure from earlier years of AI hype cycles. As Deloitte and others have noted, AI is now being treated as enterprise infrastructure, not experimental technology.
What this Means for Evidence Generation
Nowhere is this shift more visible than in evidence generation workflows.
Teams responsible for HEOR, medical affairs, and market access are under increasing pressure to produce high-quality, submission-ready evidence faster than ever—whether for Global Value Dossiers (GVDs), Joint Clinical Assessments (JCAs), or real-world evidence studies.
Embedding AI directly into these workflows enables teams to:
- Accelerate literature screening and data extraction
- Standardize methodology across projects
- Reduce manual burden while preserving expert oversight
- Deliver structured, HTA-ready outputs with greater consistency
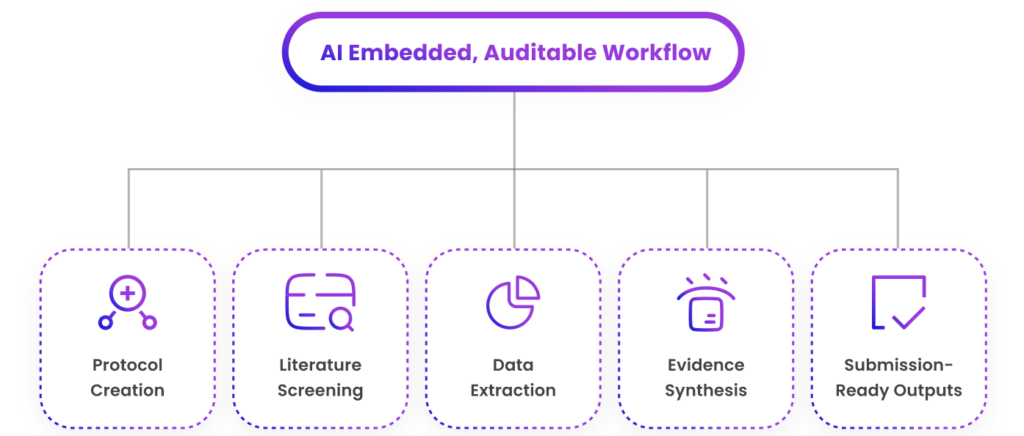 AI embedded and auditable, from protocol creation to submission-ready outputs.
AI embedded and auditable, from protocol creation to submission-ready outputs.
At MadeAi, we are already seeing organizations operationalize AI in this way, moving from one-off pilots to fully integrated, end-to-end evidence synthesis pipelines.
The Maturity Moment for AI in Life Sciences
This transition marks a fundamental shift in how the industry views AI. It is no longer a “nice-to-have” innovation; it is becoming a business-critical system.
The organizations that will lead in 2026 and beyond are those that:
- Integrate AI into their core evidence and regulatory workflows
- Establish governance frameworks that promote transparency and compliance
- Combine human expertise with AI-driven automation
- Continuously measure and optimize AI’s impact on business outcomes
How MadeAi Supports this Shift
MadeAi was designed for this exact moment, an AI-native, end-to-end platform that supports the full lifecycle of evidence generation, from protocol creation through submission-ready deliverables.
By embedding AI into the workflows that matter most, MadeAi helps life sciences teams:
- Complete literature reviews up to 60% faster
- Maintain high levels of accuracy and traceability
- Scale output without scaling headcount
- Deliver HTA- and regulatory-ready evidence packages
In a world where AI is no longer confined to sandboxes, platforms like MadeAi are enabling organizations to operationalize intelligence at scale.
Looking Ahead
Prediction #1 sets the tone for everything that follows in our 2026 outlook: AI is no longer on the sidelines; it is in the engine room of life sciences operations.
For organizations still in the pilot phase, the message is clear: The future belongs to those who move from experimentation to execution.
If you missed the 2026 Predictions for AI in Life Sciences webinar, check it out here.
AI supported the drafting of this article.






