Imagine a world where drug safety isn’t just about responding to problems after they occur, but it’s about predicting and preventing them before patients are at risk. For decades, traditional pharmacovigilance (PV) has functioned like a fire alarm system: it activates only once the smoke appears. Adverse drug reactions (ADRs) are collected through Individual Case Safety Reports (ICSRs), literature scans, and manual reviews, with signals often detected months after harm has begun.
However, as we enter 2026, this reactive model is rapidly evolving into a proactive, intelligence-driven approach powered by AI. Pharma leaders are already describing this as a fundamental shift toward “proactive risk orchestration,” where AI anticipates safety issues using real-world evidence (RWE), predictive analytics, and continuous monitoring.
This transformation is driven by surging data volumes, maturing AI technologies such as Machine Learning (ML), Natural Language Processing (NLP), and agentic systems. These advancements are supported by regulatory frameworks. Let’s explore how the industry is moving from “wait and react” to “predict and protect” and why 2026 could mark the tipping point for patient safety and operational efficiency.
Limitations of Traditional Reactive Pharmacovigilance
In the past, PV teams relied heavily on manual processes to handle millions of reports annually. Signal detection depended on human review of structured and unstructured data, often leading to delays, higher false positives, and increased regulatory risks. With global data exploding from electronic health records (EHRs), wearables, social media, and clinical trials, the old system simply couldn’t keep pace.
Visual Comparison: Reactive vs. Proactive Pharmacovigilance
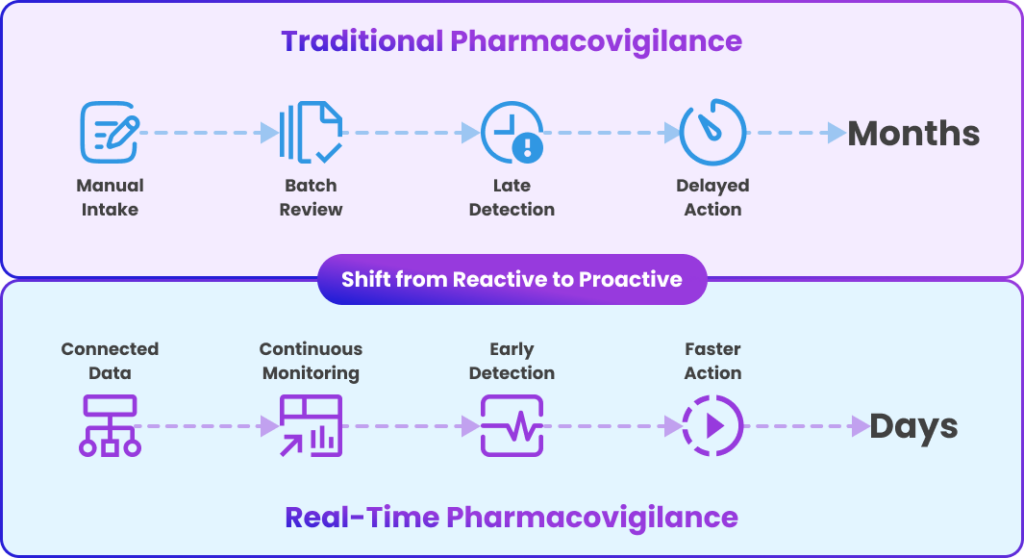 From reactive to proactive pharmacovigilance
From reactive to proactive pharmacovigilance
As this infographic shows, traditional methods often take months for signal detection through slow, manual workflows. In contrast, AI-enabled systems deliver insights in days via real-time, continuous monitoring by dramatically improving response times.
How AI Powers the Proactive Revolution
AI is transforming pharmacovigilance by shifting the approach from a reactive to a proactive model. AI is now enabling continuous monitoring, analyzing data from a variety of sources, including clinical trials, medical records, and social media, to identify potential risks before they fully emerge. By leveraging advanced algorithms, AI can detect patterns and early signals in real time, which allows safety teams to act much sooner and with greater precision. This proactive approach enhances patient safety, speeds up the decision-making process, and helps ensure regulatory compliance.
Despite its significant capabilities, AI doesn’t replace the need for human expertise. Instead, it augments the work of pharmacovigilance professionals by offering actionable insights that inform decision-making. The integration of AI allows experts to focus on more complex tasks such as evaluating the severity of risks, understanding the context, and communicating findings to regulatory bodies. Together, AI and human judgment form a powerful synergy that is reshaping pharmacovigilance into a more efficient, agile, and accountable discipline. This shift represents a crucial leap in how pharmaceutical companies protect patients while staying ahead of evolving regulatory requirements.
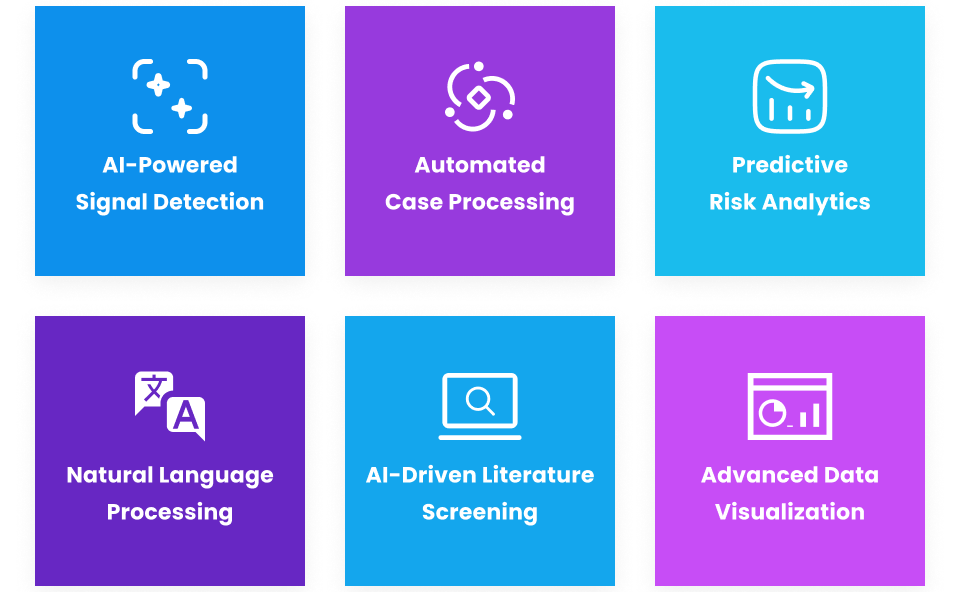 End-to-end AI for drug safety
End-to-end AI for drug safety
This visual highlights key applications, including automated case processing, real-time signal detection, predictive analytics, and multi-source data integration.
AI Touchpoints Across the PV Lifecycle
In the next phase of pharmacovigilance, AI becomes the connective intelligence across the entire lifecycle, managing workflows from adverse event intake to signal detection and regulatory reporting. Every touchpoint evolves into a living, responsive system where data flows continuously rather than in isolated steps. Case narratives are structured in real time, duplicates are resolved instantly, and emerging safety signals are surfaced with contextual evidence layered in. Human experts remain embedded at critical checkpoints, validating, interpreting, and guiding decisions where judgment, accountability, and regulatory responsibility matter most.
What emerges is a hybrid model where AI drives speed, consistency, and traceability, while humans anchor trust and compliance. Safety teams move from reactive processing to proactive oversight, supported by AI that acts as a second reviewer across every stage. Decisions are no longer delayed by manual bottlenecks but are accelerated through governed workflows designed for transparency and audit readiness. In this future, pharmacovigilance is not just faster. It is continuously learning, deeply integrated, and built on a foundation where AI and human expertise operate in deliberate alignment.
Transitioning smoothly toward real-world impact, companies are already piloting these capabilities to move safety upstream into clinical development.
2026 Predictions: What Proactive PV Will Look Like
By the end of 2026, several key advancements are expected to become mainstream:
Real-time intelligent surveillance: Continuous monitoring of RWE, wearables, and multi-source data will flag signals in hours rather than months.
Predictive risk orchestration: AI will simulate outcomes and recommend preemptive actions, shifting PV from post-market surveillance to translational safety.
Self-optimizing agentic workflows: Autonomous systems will handle routine tasks like report drafting while adapting dynamically to regulatory demands.
Personalized safety insights: Tailored predictions for individual patient profiles, supported by inclusive real-world data.
Here’s a side-by-side comparison for clarity:
| Aspect | Reactive PV (Traditional) | Proactive PV with AI (2026 Outlook) |
|---|---|---|
| Signal Detection | Manual, post-report review | AI-powered, real-time across RWE, literature & wearables |
| Risk Management | Mitigation after events | Predictive modeling & preemptive interventions |
| Data Sources | Primarily ICSRs | Multi-source: EHRs, social media, genetics |
| Response Time | Weeks to months | Hours to days |
| Human Role | Heavy manual processing | Strategic oversight & clinical judgment |
| Patient Impact | Delayed intervention risks | Earlier protection & personalized safety insights |
These predictions align closely with insights from industry experts and perspectives on proactive drug safety.
Regulatory Support and Governance
Regulators are actively enabling this shift. The FDA’s Emerging Drug Safety Technology Program (EDSTP) serves as a key forum for collaboration on AI in pharmacovigilance. In January 2026, the FDA and EMA jointly released the Guiding Principles of Good AI Practice in Drug Development, emphasizing human-centric design, transparency, risk-based approaches, and robust governance.
These frameworks ensure AI tools remain explainable and compliant, addressing common concerns around data bias and “black box” models.
Benefits, Challenges, and Preparation Steps
The benefits are compelling: fewer ADRs, faster approvals, cost savings, and ultimately safer medicines for patients worldwide. Yet challenges persist, including ensuring high-quality data foundations, building explainable models, and maintaining strong human-AI collaboration.
To prepare, organizations should:
- Audit and harmonize their data ecosystems.
- Pilot AI for high-volume tasks like case intake and signal detection.
- Invest in upskilling teams and establishing clear governance frameworks.
The Bottomline
The shift from reactive to proactive pharmacovigilance isn’t about replacing safety scientists with machines. It’s about equipping experts with better tools, that is, ones that can process more data, in more languages, and at faster speeds than any human team could manage on its own. The expert still makes the key decisions: interpreting what a signal means, deciding how to communicate it to regulators, and determining the necessary actions.
What changes is the focus of the questions. Instead of asking, “Did enough reports come in for us to notice this?”, the new question becomes: “Our models flagged this three weeks ago. Here’s the evidence, the patient risk profile, and what the mechanism literature says. What do you think?” This approach fosters a much more meaningful conversation about drug safety. Building the infrastructure to make this process reliable, scalable, and compliant with evolving regulations is the big engineering challenge of this decade.
Author’s Note: This article was supported by AI-based research and writing, with Claude 4.6 assisting in the creation of text and images.
FAQs
What is proactive pharmacovigilance, and how does it differ from the traditional reactive approach?
Proactive pharmacovigilance leverages AI and real-world data to predict and prevent adverse events before they occur, moving beyond the traditional model that only responds after reports are filed.
How is AI expected to transform pharmacovigilance by 2026?
AI enables faster, more accurate signal detection, predictive risk modeling, and automated workflows, shifting the field toward real-time intelligent surveillance.
Will AI replace human professionals in pharmacovigilance?
No, AI augments expertise by handling repetitive tasks, allowing safety scientists to focus on clinical interpretation, strategic decisions, and regulatory oversight.
What are the key benefits of adopting AI for proactive pharmacovigilance?
Benefits include earlier risk identification (reducing ADRs), workload reductions of up to 80%, cost efficiencies, improved compliance, and enhanced patient safety outcomes.
What regulatory guidelines support AI use in pharmacovigilance?
The FDA’s EDSTP and the joint FDA-EMA Guiding Principles (2026) provide frameworks for transparent, human-centric, and compliant AI deployment.
How can pharmaceutical companies in India and globally prepare for proactive PV in 2026?
Start with data quality improvements, pilot AI tools for signal detection, develop governance policies, and train teams on hybrid human-AI workflows. (Relevant for readers in Tamil Nadu, India, and beyond.)
What are the main challenges in implementing AI-driven pharmacovigilance?
Key hurdles include data bias, model explainability, legacy system integration, and ensuring robust governance to maintain regulatory trust.




