For years, the pharmaceutical industry has followed a familiar cycle: design a clinical trial, execute it, lock the database, analyze the results, and then wait for the next study to answer remaining questions. Wait for post-market surveillance to show real-world outcomes. Wait for regulators or payers to request more data before approving reimbursement. However, this long-standing rhythm is beginning to change in 2026.
Questions multiply faster than discrete studies can answer them. Precision medicine requires evidence for specific patient groups. Health technology assessment bodies seek comparative effectiveness across pathways. Regulators require continuous safety monitoring throughout a product’s life. Patients want to know if a therapy suits someone like them, not just an average from years-old trials. Therefore, our fourth prediction stands clear: by the end of 2026, how we generate evidence in pharmaceutical development will change from isolated events to continuous evidence generation. This means agent-led processes are integrated into clinical research and routine care.
The Growing Evidence Gap in Traditional Approaches
Traditional clinical trials deliver high-quality evidence, yet they remain limited. A Phase III trial enrolls selected patients, measures predefined endpoints, and captures a snapshot of efficacy and safety. This informs approval, but stakeholders need much more.
Providers ask how therapies perform in comorbid patients excluded from trials. Payers seek comparative data against alternatives for coverage. HTA bodies require real-world cost-effectiveness proof. Precision medicine further fragments needs. Different biomarkers, genetics, or stages each require validation.
Statistics highlight the inefficiency. Only about 20% of leading companies create integrated lifecycle evidence plans. Trials often need amendments (76% of Phase I-IV studies), adding months and high costs. We generate evidence too slowly, for too narrow groups, and miss many real-world questions.
How Agentic AI Enables Continuous Evidence Generation
Agentic AI shifts evidence development from reactive to proactive. Unlike earlier tools that automated single tasks, these systems perceive context, reason through scenarios, plan workflows, and act toward goals with limited human input.
In evidence contexts, agents continuously monitor real-world streams, spot emerging gaps, design studies to address them, orchestrate multi-source data collection, synthesize insights, and deliver tailored findings. Often, they anticipate stakeholder needs.
Three capabilities set 2026 systems apart:
- Autonomous study design and optimization: Agents review literature, analyze competitors, assess site feasibility, and propose optimized protocols based on real-world likelihood of success.
- Continuous real-world evidence synthesis: Agents watch EHRs, claims, registries, and wearables in real time, flagging patterns like safety signals or new efficacy subgroups to trigger deeper analysis.
- Adaptive evidence delivery: Living repositories generate on-demand, audience-specific analytics, updating models and scenarios instantly for HTA requests or payer queries.
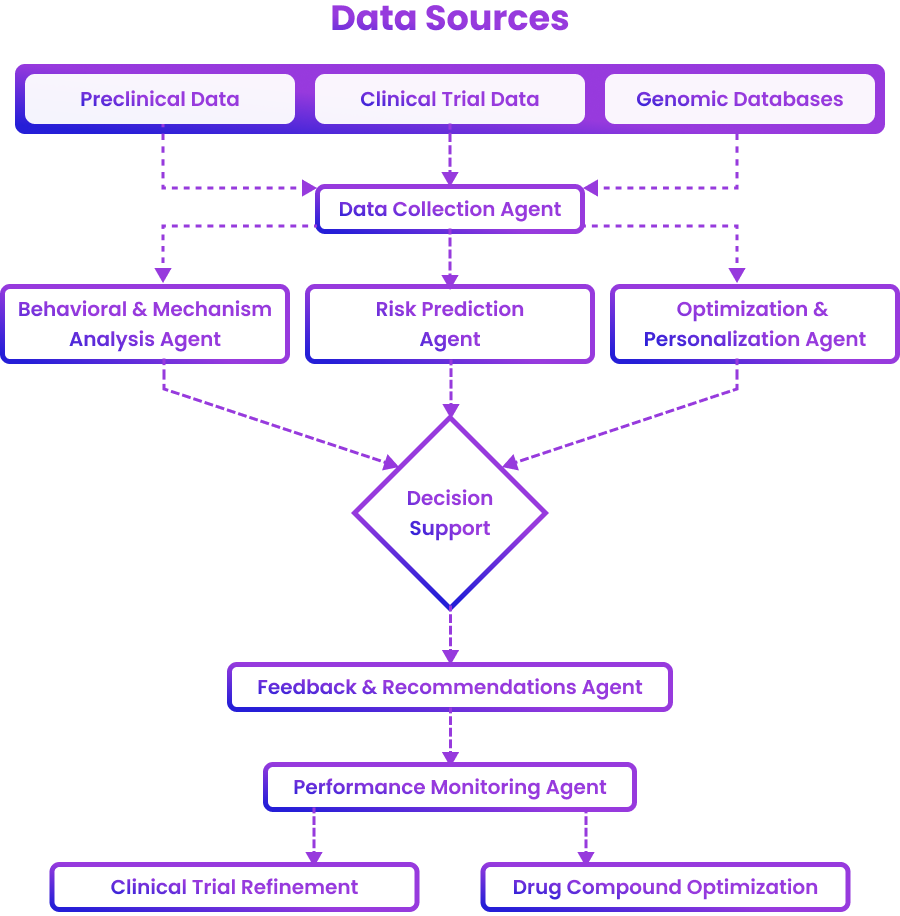 How agentic AI connects data, analysis, and decisions for continuous evidence generation
How agentic AI connects data, analysis, and decisions for continuous evidence generation
Image Source: Adapted from an external source for illustrative purposes.
From Episodes to Ecosystems: The Architecture Shift in Continuous Evidence Generation
Traditional evidence follows isolated episodes: plan, execute, analyze, publish, archive. Each study stands alone.
Continuous evidence generation demands a living ecosystem architecture. Organizations in 2026 converge on key patterns.
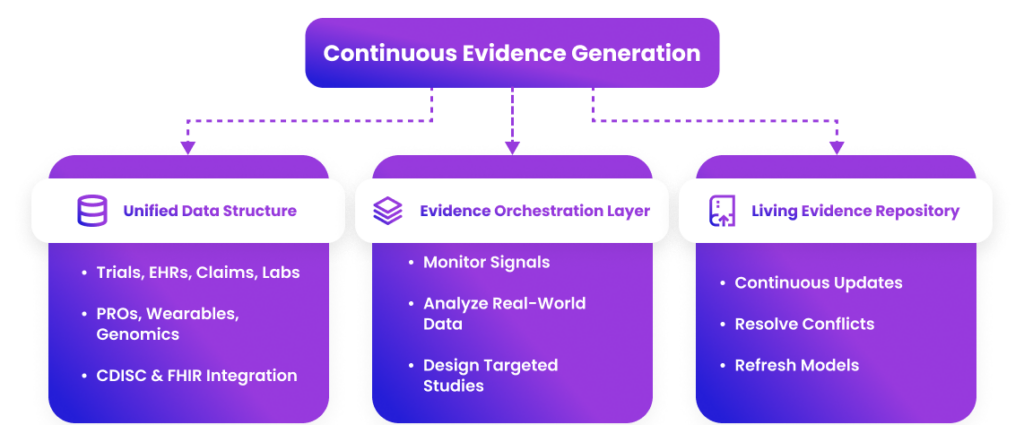 Architecture of continuous evidence generation with unified data and orchestration
Architecture of continuous evidence generation with unified data and orchestration
Traditional vs. Continuous Evidence Generation
| Dimension | Traditional Episodic Model | Continuous Agent-Led Model |
|---|---|---|
| Study Initiation | Months of planning before the first patient | Agents identify gaps and propose studies in days |
| Data Sources | Single source per study, fragmented databases | Unified fabric integrating trial, RWE, registry data |
| Evidence Updates | New study for each question | Living knowledge graphs updated as data arrives |
| Timeline | 12-36 months from question to answer | Days to weeks for insights; ongoing refinement |
| Stakeholder Delivery | Static retrospective reports | On-demand tailored analytics |
| Adaptability | Fixed protocols; costly amendments | Agents adjust strategies based on emerging patterns |
Real-World Implementations in 2026
The shift from theoretical possibility to operational reality is happening now. Multiple pharmaceutical organizations and technology providers launched production systems in early 2026 that demonstrate what continuous evidence generation looks like in practice.
MadeAi’s AI-Powered Evidence Synthesis Platform
MadeAi-LR streamlines the evidence synthesis for HEOR, Medical Affairs, Market Access, and RWE in life sciences. It enables teams to scale operations, reduces timelines, and delivers high-quality services across the full evidence synthesis lifecycle, from protocol development and smart literature search through AI-assisted screening, data extraction, summarization, and final report authoring.
Recursion’s ClinTech Initiative
AI-driven drug developer Recursion has deployed agentic systems focused on three pillars: smarter trial design, accelerated enrollment, and enhanced evidence generation. Their agents continuously analyze internal compound libraries, published literature, competitive trial data, and real-world patient populations to identify optimal study parameters before human teams finalize protocols. The system has reduced protocol amendment rates by identifying mismatches between design assumptions and operational realities before trials launch.
ConcertAI’s Accelerated Clinical Trials Platform
Launched at SCOPE 2026, specialized agents handle literature scanning, protocol design, feasibility, and real-time patient matching from EHRs. Reports show up to 50% reductions in design timelines and amendments.
IQVIA’s Agentic Trial Workflows
IQVIA’s implementation focuses on the operational bottlenecks that plague trial execution, including site activation, startup activities, and data review setup. Agents automate administrative gaps in site activation, startup, and data review, freeing teams for safety and quality focus.
Bridging Clinical Research and Routine Care
Agentic systems help research-embedded care to become a practical reality. Trials integrate into EHRs, workflows, and ordering processes, reducing burden and generating immediately applicable evidence from real patterns.
Recent publications by the FDA’s Real-World Evidence Framework and ICH E6(R3) Good Clinical Practice Guideline emphasize unified ecosystems with methodological safeguards for routine-care evidence.
The infrastructure requirements extend beyond the technical aspects. Health systems must value trials in care, payments support longitudinal collection, and IRBs streamline pragmatic designs. Culture change matters as much as technical capability.
Trust, Transparency, and the Human Element
Autonomous evidence generation raises fundamental questions about trust and oversight. When AI agents design studies, recruit patients, monitor safety signals, and synthesize findings with minimal human intervention, how do we ensure scientific rigor? How do we maintain accountability? How do we prevent automation from introducing systematic biases that human reviewers might catch?
The principle emerging as best practice: automation should augment human expertise, not replace it. The most effective systems combine both, with clear delineation of where the machine stops and the human begins.
Data quality remains paramount, and agentic systems are only as good as the data they consume. Incomplete electronic health records, inconsistent coding practices, fragmented data across systems, and biased datasets all propagate into agent-generated evidence. Organizations investing in continuous evidence generation must invest equally in data infrastructure, standardization, and governance.
Measuring Impact: Early Adopter Results
Early adopters of agent-led evidence generation are reporting measurable improvements across multiple dimensions:
- Timeline Compression: Study design timelines reduced 25-50%
- Cost Reduction: Protocol amendments down ~50%, saving costs and time
- Enrollment Acceleration: Recruitment accelerated 25-50% with better diversity
- Evidence Spread: Broader evidence answering multiple questions from unified data
- Operational Efficiency: CRAs redirect 30-40% time from admin to oversight
Clinical outcomes matter most: better decisions, precise matching, faster signal detection.
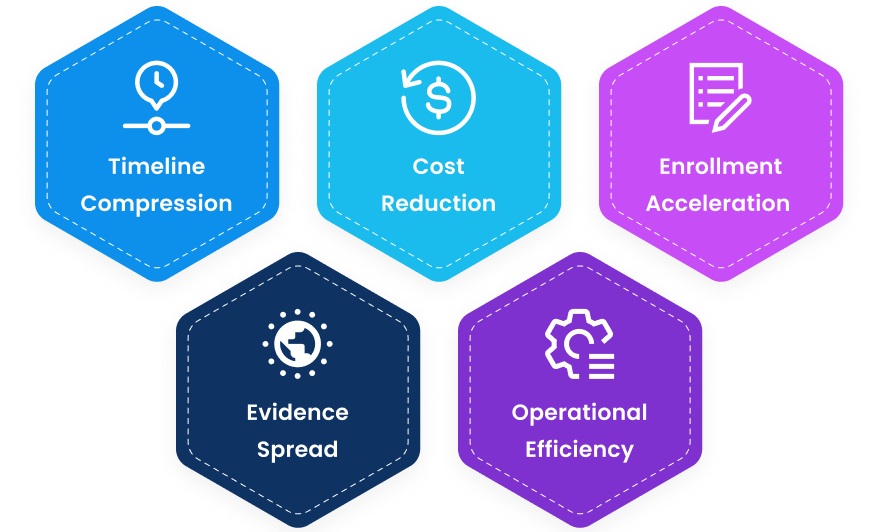 Key impact areas of agent-led evidence generation
Key impact areas of agent-led evidence generation
The Regulatory and Reimbursement Landscape
Regulators are moving forward carefully. The FDA supports the use of real-world evidence, while the EMA and MHRA are building clear guidelines. Transparency is essential, and teams need to show how AI works, what data it uses, and its limits. Living documentation helps keep compliance up to date in real time.
HTA bodies face format challenges but gain from dynamic analytics. Collaboration refines requirements.
The Future: The 2027 Evidence Landscape
By late 2027, continuous evidence generation will become standard for leaders. Advantages include faster access, stronger payer ties, improved outcomes, and lower costs.
Technology matures: better reasoning, uncertainty handling, multi-stakeholder optimization. Data interoperability advances. Regulations clarify AI evidence expectations.
Cultural shifts redefine roles. Experts curate graphs, monitor agents, interpret insights, and guide strategy. Work focuses on the right questions and wise application.
The Evidence Revolution
We’re witnessing the transformation of evidence generation from a bottleneck in pharmaceutical development to a continuous flow that keeps pace with the questions we need to answer. Agentic AI makes this possible by automating the orchestration of complex, multi-source data synthesis while maintaining the rigor that regulatory decisions and patient safety demand.
Leaders integrate generation into research and care, build trust via transparency, and use insights for faster decisions. In 2026, evidence becomes continuous, adaptive, comprehensive, and integrated, not consulted retrospectively. The pharmaceutical industry has needed this transformation for decades. The technology, the regulatory environment, and the competitive pressures have finally aligned to make it possible.
Key Takeaways
- Traditional trials provide snapshots, missing many stakeholder questions.
- Agentic AI enables autonomous design, real-world synthesis, and tailored delivery in real time.
- 2026 implementations cut timelines and amendments by 25-50%.
- Research-care integration yields real-world, diverse evidence.
- Success demands guardrails: oversight, audits, and quality validation.
Author’s Note: This article was supported by AI-based research and writing, with Claude 4.5 assisting in the creation of text and images.
FAQs
What is continuous evidence generation?
It is the shift from episodic clinical trials to ongoing, agent-led processes that collect, synthesize, and deliver evidence in real time, integrating trial data with real-world sources for faster, broader insights.
Why are traditional trials no longer sufficient?
Trials produce limited snapshots for selected populations and predefined endpoints. They cannot quickly address emerging questions from precision medicine, payers, HTA bodies, or real-world use, leading to delays, high costs, and narrow applicability.
How do agentic AI systems support this shift?
Agents autonomously monitor data streams, identify gaps, design optimized studies, orchestrate multi-source collection, and generate tailored, on-demand analytics, often proactively before stakeholders request it.
What are the main architectural components?
A unified data fabric for seamless access, an orchestration layer for coordinated agent teams, and living knowledge graphs that dynamically update evidence repositories and downstream models.
What guardrails ensure trust and quality?
Human oversight on critical decisions, bounded agent autonomy, comprehensive audit logs, bias checks, and validation metrics that confirm agents improve evidence quality, not just speed.
What measurable benefits have early adopters seen?
Reductions of 25-50% in design and recruitment timelines, ~50% fewer protocol amendments, 30-40% less administrative time for CRAs, and broader evidence addressing multiple stakeholder needs from unified data.





