GenAI-enabled Global Value Dossiers
Navigating market access requires compelling, evidence-rich Global Value Dossiers (GVDs) that are accurate, adaptive, and aligned with payer expectations. MadeAi-GVD streamlines the end-to-end development of both traditional and interactive GVDs—leveraging GenAI and domain expertise to reduce manual burden, accelerate timelines, and ensure local adaptability.
Whether you're preparing for HTA submissions, AMCP dossiers, or global affiliate support, MadeAi offers an intelligent, transparent solution for building modular, customizable, and audit-ready dossiers at scale.
Lorem ipsum dummy text
Why Choose MadeAi for GVD?
- Up to 50% faster GVD creation cycles
- Full transparency and source traceability
- Scalable support for multiple therapeutic areas
- Built-in alignment with QALY, RWE, and HTA standards
- Proven by top-15 pharma and global biotech innovators
Why Choose MadeAi for GVD?
- Up to 50% faster GVD creation cycles
- Full transparency and source traceability
- Scalable support for multiple therapeutic areas
- Built-in alignment with QALY, RWE, and HTA standards
- Proven by top-15 pharma and global biotech innovators

What MadeAi Delivers for GVDs
MadeAi streamlines Global Value Dossier development by accelerating evidence synthesis, ensuring full source traceability, and aligning with HTA, QALY, and RWE standards. Its GenAI-powered platform supports multiple therapeutic areas with scalable precision, reducing GVD creation cycles by up to 50%. Built for compliance and speed, MadeAi is trusted by leading pharma and biotech teams to deliver audit-ready, insight-rich dossiers at scale.
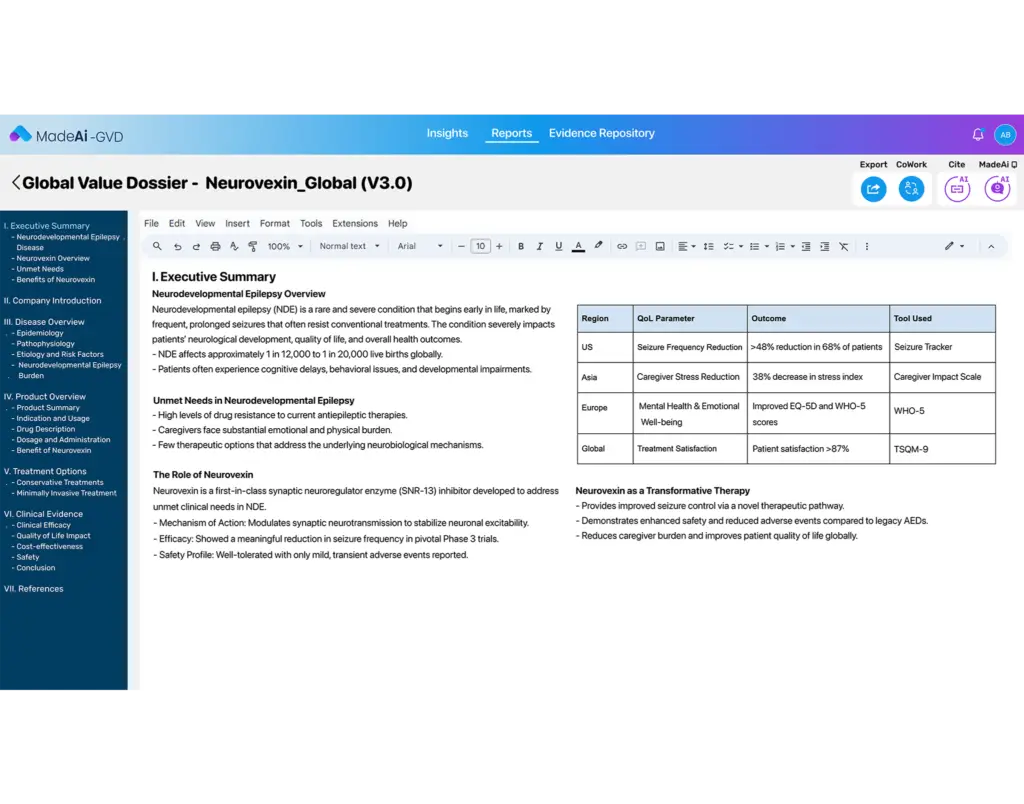
End-to-End Dossier Development
From disease burden analysis to modeling and market access strategy, MadeAi helps teams assemble comprehensive and compliant dossiers with speed and precision.
AI-Assisted Content Generation
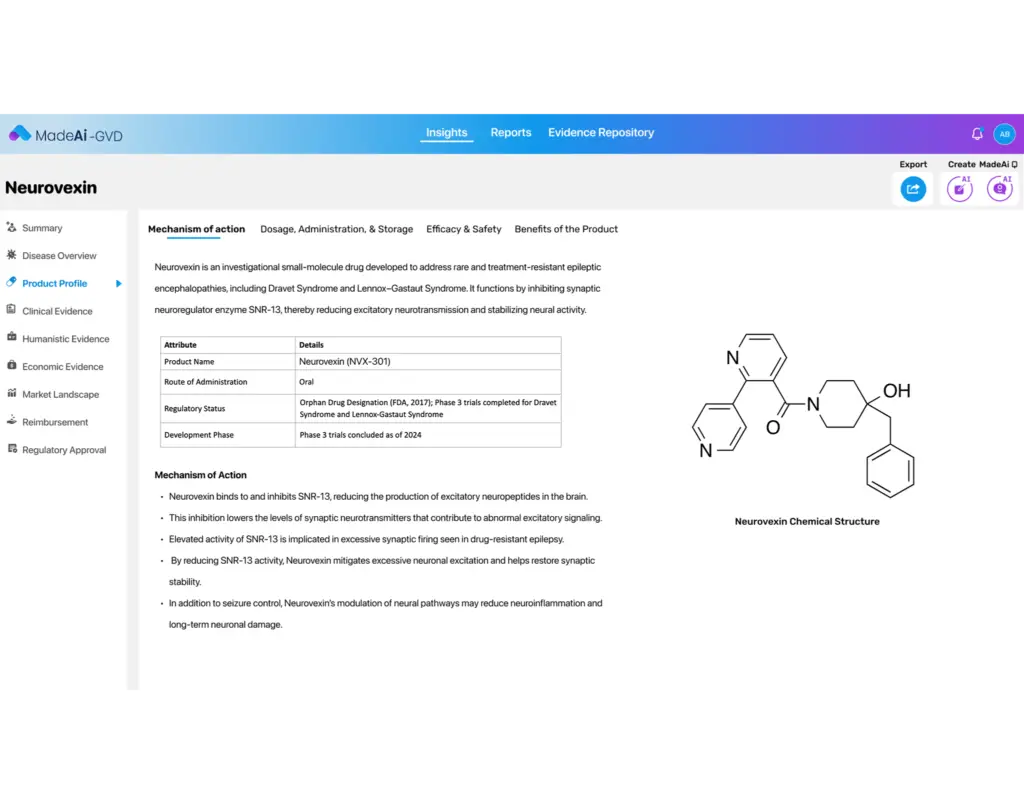
Automatically draft core sections like value stories, unmet need narratives, and product positioning using AI trained on medical, regulatory, and payer-relevant content.
Interactive GVD (iGVD) Options
Support for dynamic, stakeholder-specific digital dossiers that include visualization tools, slide generators, and adaptive navigation for cross-functional use.


Integrated Compliance Frameworks
Built-in checks and references for HTA and AMCP standards to ensure submission-ready outputs and local market adaptation needs.
Collaboration and Version Control
Assign roles to medical, HEOR, regulatory, marketing, and legal teams with centralized input tracking, version history, and modular content updates.
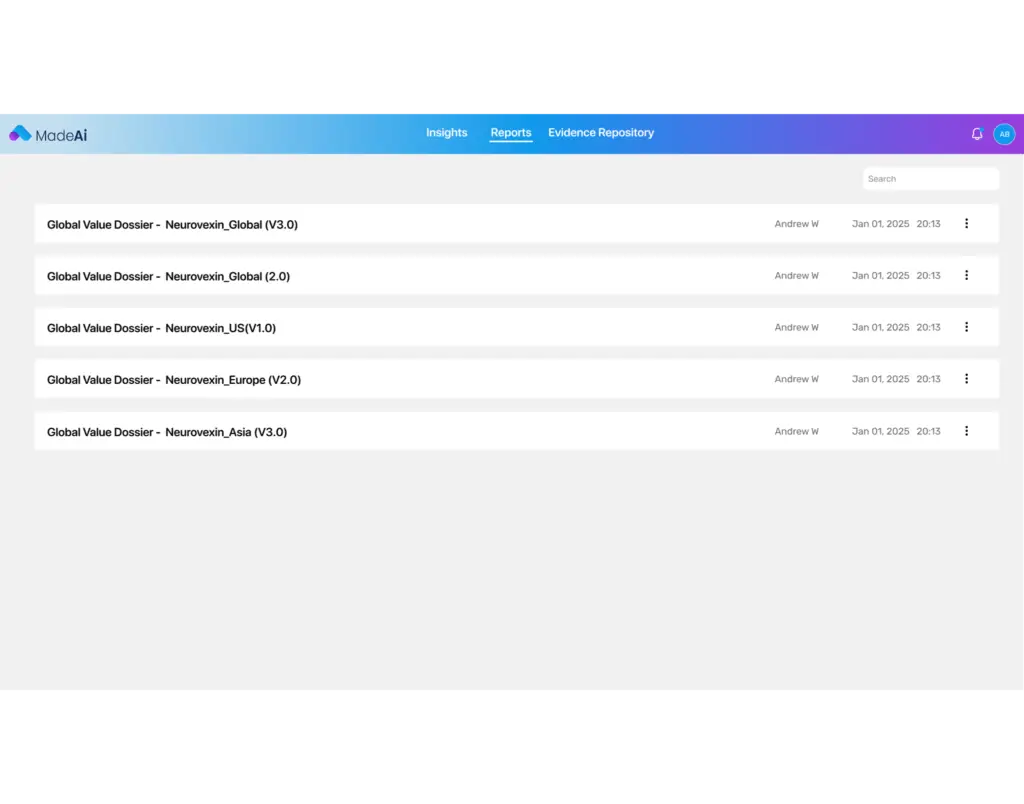

Searchability & Access
With MadeAi’s GenAI-enabled InsightsBot, information with in GVDs becomes simple search and access, empowering teams to find critical data and documents quickly freeing up resource time.
Living GVD
The emergence of new clinical data, regulatory changes, and market access updates are easily managed with MadeAi’s Living GVD, which automatically monitors for and incorporates relevant market events to keep GVD’s up to date.
Who It’s For
- HEOR & Market Access Teams:
Deliver high-quality dossiers across global markets - Regulatory & Compliance Teams:
Ensure traceability and alignment with agency requirements - Medical Affairs:
Communicate differentiated clinical value clearly and efficiently - Launch Teams:
Scale dossier creation across multiple product launches


Contact Info
Connect with a MadeAi representative to see how we can support your GVD development needs.
FAQs
What is a Global Value Dossier (GVD), and why is it important?
A GVD is a comprehensive document that presents evidence about a product’s value for payers, HTA agencies, and market access teams. It’s crucial because a strong GVD enables informed decisions and successful healthcare product launches across markets.
How does MadeAi leverage GenAI to improve GVD development?
MadeAi’s platform uses advanced AI trained on medical, regulatory, and payer-relevant content to automate the drafting of core dossier sections, accelerating evidence synthesis and narrative creation while reducing manual effort.
How much faster can MadeAi complete GVD cycles compared to traditional approaches?
MadeAi claims up to a 50% reduction in GVD development cycles by streamlining key steps with AI-powered automation and modular workflows and integrated compliance frameworks. This proven acceleration helps teams meet tight launch timelines while maintaining quality.
Can the platform support dossiers for multiple therapeutic areas?
Yes, MadeAi is designed for scalability and can be used for GVDs in diverse therapeutic areas, making it valuable for organizations with broad product portfolios. The platform adapts to different disease contexts, making it valuable for organizations with multiple products and varied market access needs.
What distinguishes MadeAi’s Interactive GVD (iGVD) from traditional dossiers?
The iGVD delivers dynamic, stakeholder-specific digital experiences that go beyond static documents. Features include visualization tools, slide generators, and adaptive navigation that enable cross-functional collaboration and tailored communication with different audiences—from payers to internal leadership.
How does MadeAi ensure compliance with global standards and local adaptability?
MadeAi incorporates built-in compliance checks and references aligned with QALY, RWE, HTA, and AMCP standards. From disease burden analysis to modeling and market access strategy, the platform ensures dossiers are audit-ready, submission-compliant, and adaptable to specific local market requirements across regions.
Can MadeAi help manage frequent updates resulting from new clinical or regulatory data?
Absolutely. The Living GVD feature actively monitors for and automatically incorporates new clinical data, regulatory changes, and market access updates. This keeps documents current without requiring manual overhauls, ensuring your dossiers reflect the latest evidence and market dynamics.
Does the platform provide transparency and source traceability?
Yes, full source traceability is a core feature embedded throughout the platform. Users can review and verify the evidence supporting each dossier section, enhancing both quality control and compliance with agency requirements. This transparency is critical for regulatory defense and stakeholder confidence.
Is collaboration across medical, HEOR, regulatory, marketing, and legal teams supported?
MadeAi includes robust integrated collaboration tools that allow different teams to work seamlessly together. You can assign specific roles, track centralized input, manage version history, and coordinate modular content updates efficiently, ensuring all stakeholders stay aligned throughout development.
How does MadeAi’s InsightsBot improve information access within dossiers?
InsightsBot is a GenAI-enabled search tool that allows users to quickly find and retrieve critical data or documents from within GVD repositories. This simple search and access functionality dramatically reduces time spent on information retrieval, freeing up resources and enhancing team productivity.
What kind of organizations can benefit most from MadeAi’s GVD solution?
MadeAi is ideal for pharmaceutical and biotech companies that need audit-ready dossiers for multiple products and markets. The platform is proven by top-15 pharma and global biotech innovators, benefiting:
- HEOR & Market Access Teams: Delivering high-quality dossiers across global markets
- Regulatory & Compliance Teams: Ensuring traceability and agency alignment
- Medical Affairs: Communicating differentiated clinical value clearly and efficiently
Launch Teams: Scaling dossier creation across multiple product launches
Can the platform help with both traditional and digital (interactive) dossier formats?
Yes, MadeAi supports end-to-end development of both static and dynamic GVDs. Whether you need traditional submission documents for HTA agencies or interactive digital dossiers for internal reviews and stakeholder engagement, the platform flexibly accommodates various use cases and audience needs.
What standards and frameworks does MadeAi align with by default?
The platform aligns with Quality-Adjusted Life Years (QALY), Real-World Evidence (RWE), Health Technology Assessment (HTA), and AMCP standards, covering major global regulatory requirements.
How does MadeAi address data security and confidentiality?
MadeAi is built as a compliance-oriented SaaS platform designed for life sciences, ensuring audit-ready outputs and rigorous security protocols. The platform’s focus on source traceability, version control, and compliance frameworks reflects its commitment to handling sensitive pharmaceutical data with appropriate confidentiality and security measures.
Who should I contact to discuss implementation or request a demo?
You can connect directly with a MadeAi representative through the website’s contact information to discuss your organization’s GVD needs or request a tailored demonstration.



